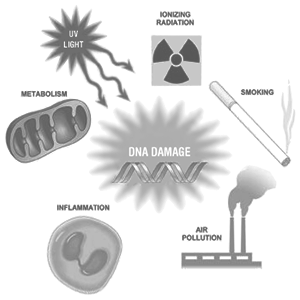
The free radical theory of aging states that organisms age because cells accumulate free radical damage over time. A free radical is a molecule with an unpaired electron. The molecule is reactive and seeks another electron to pair. This initiates an uncontrolled chain reaction that can damage the natural function of the living cell, causing various diseases. While a few free radicals such as melanin are not chemically reactive, most biologically-relevant free radicals are highly reactive.
For most biological structures, free radical damage is closely associated with oxidative damage. Antioxidants are reducing agents, they limit oxidative damage to biological structures by donating an electron to free radicals. Biogerontologist Denham Harman first proposed the free radical theory of aging in the 1950s, and in the 1970s extended the idea to implicate mitochondrial production of reactive oxygen species.
In some model organisms, such as yeast and Drosophila (fruit fly), there is evidence that reducing oxidative damage can extend lifespan. In mice, interventions that enhance oxidative damage generally shorten lifespan. However, in roundworms, blocking the production of the naturally occurring antioxidant superoxide dismutase has recently been shown to increase lifespan. Whether reducing oxidative damage below normal levels is sufficient to extend lifespan remains an open and controversial question.
When the free radical theory of aging was first proposed the prevailing scientific opinion held that free radicals were too unstable to exist in biological systems. This was also before anyone invoked free radicals as a cause of degenerative diseases. Two sources inspired Harman: 1) the rate of living theory, which holds that lifespan is an inverse function of metabolic rate which in turn is proportional to oxygen consumption, and 2) Argentine biologist Rebbeca Gershman’s observation that hyperbaric (high pressure) oxygen toxicity and radiation toxicity could be explained by the same underlying phenomenon: oxygen free radicals. Noting that radiation causes ‘mutation, cancer and aging,’ Harman argued that oxygen free radicals produced during normal respiration would cause cumulative damage which would eventually lead to organismal loss of functionality, and ultimately death.
In later years, the free radical theory was expanded to include not only aging per se, but also age-related diseases. Free radical damage within cells has been linked to a range of disorders including cancer, arthritis, atherosclerosis, Alzheimer’s disease, and diabetes. There has been some evidence to suggest that free radicals and some reactive nitrogen species trigger and increase cell death mechanisms within the body such as apoptosis (cell self-destruction) and in extreme cases necrosis (cell death). In 1972, Harman modified his original theory to what became known as the ‘mitochondrial theory of aging.’ In its current form, this theory proposes that reactive oxygen species that are produced in the mitochondria, causes damage to certain macromolecules including lipids, proteins, and most importantly mitochondrial DNA. This damage then causes mutations which leads to an increase of ROS (reactive oxygen species, chemically reactive molecules containing oxygen) production and greatly enhances the accumulation of free radicals within cells. Since Harman first proposed the free radical theory of aging, there have been continual modifications and extensions.
Free radicals are atoms or molecules containing unpaired electrons. Electrons normally exist in pairs in specific orbitals in atoms or molecules. Free radicals, which contain only a single electron in such any orbital, are usually unstable toward losing or picking up an extra electron, so that all electrons in the atom or molecule will be paired. Damage occurs when the free radical encounters another molecule and seeks to find another electron to pair its unpaired electron. The free radical often pulls an electron off a neighboring molecule, causing the affected molecule to become a free radical itself. The new free radical can then pull an electron off the next molecule, and a chemical chain reaction of radical production occurs. The free radicals produced in such reactions often terminate by removing an electron from a molecule which becomes changed or cannot function without it, especially in biology. Such an event causes damage to the molecule, and thus to the cell that contains it (since the molecule often becomes dysfunctional).
The chain reaction caused by free radicals can lead to cross-linking of atomic structures. In cases where the free radical-induced chain reaction involves base pair molecules in a strand of DNA, the DNA can become cross-linked. DNA cross-linking can in turn lead to various effects of aging, especially cancer. Other cross-linking can occur between fat and protein molecules, which leads to wrinkles. Free radicals can oxidize LDL (low density lipoproteins, or ‘bad cholestero’l), and this is a key event in the formation of plaque in arteries, leading to heart disease and stroke. These examples are examples of how the free-radical theory of aging has been used to neatly ‘explain’ the origin of many chronic diseases.
Antioxidants are helpful in reducing (donating electrons) and preventing damage from free radical reactions because of their ability to neutralize the radical without forming another. Ascorbic acid (Vitamin C), for example, can lose an electron to a free radical and remain stable itself by passing its unstable electron around the antioxidant molecule. This has led to the hypothesis that large amounts of antioxidants, with their ability to decrease the numbers of free radicals, might lessen the radical damage causing chronic diseases, and even radical damage responsible for aging. However, some antioxidant supplements may promote disease and increase mortality in humans. Free radicals induce an endogenous response which protects against exogenous radicals (and possibly other toxic compounds). Recent experimental evidence strongly suggests that this is indeed the case, and that such induction of endogenous free radical production extends the life span of roundworms.
Most importantly, this induction of life span is prevented by antioxidants, providing direct evidence that toxic radicals may mitohormetically exert life extending and health promoting effects. ROS may perform an essential and potentially lifespan-promoting role as redox signaling molecules which transduce signals from the mitochondrial compartment to other compartments of the cell. Increased formation of reactive oxygen species (ROS) within the mitochondria may cause an adaptive reaction which produces increased stress resistance and a long-term reduction of oxidative stress. This kind of reverse effect of the response to ROS stress has been named mitochondrial hormesis or mitohormesis and is hypothesized to be responsible for the respective lifespan-extending and health-promoting capabilities of glucose restriction and physical exercise.
Hormesis may also be induced by endogenously produced, potentially toxic agents. For example, mitochondria consume oxygen which generates free radicals (reactive oxygen species) as an inevitable by-product. It was previously proposed on a hypothetical basis that such free radicals may induce an endogenous response culminating in increased defense capacity against exogenous radicals (and possibly other toxic compounds). Recent experimental evidence strongly suggests that this is indeed the case, and that such induction of endogenous free radical production extends life span of a model organism. Most importantly, this extension of life span is prevented by antioxidants, providing direct evidence that toxic radicals may mitohormetically exert life extending and health promoting effects. Since mitochondrial activity was found to be increased in the before-mentioned studies, this effect cannot be explained by an excess of free radicals that might mark mitochondria for destruction by lysosomes, with the free radicals acting as a signal within the cell to indicate which mitochondria are ready for destruction, as proposed by British biochemist Nick Lane.
Studies have demonstrated that calorie restriction displays positive effects on the lifespan of organisms even though it is accompanied by increases in oxidative stress. Many studies suggest this may be due to anti-oxidative action, oxidative stress suppression, or oxidative stress resistance which occurs in calorie restriction. Fontana et al. suggest calorie restriction influenced numerous signal pathways through the reduction of insulin-like growth factor I (IGF-1). The increase in life expectancy observed during some calorie restriction studies which can occur with lack of decreases or even increases in O2 consumption is often inferred as opposing the mitochondrial free radical theory of aging. However, Barja showed significant decreases in mitochondrial oxygen radical production (per unit of O2 consumed) occur during dietary restriction, aerobic exercise, chronic exercise, and hyperthyroidism. Additionally, mitochondrial oxygen radical generation is lower in long-lived birds than in short-lived mammals of comparable body size and metabolic rate. Thus, mitochondrial ROS production must be regulated independently of O2 consumption in a variety of species, tissues and physiologic states.
The free radical theory of aging implies that antioxidants such as vitamin A, vitamin C, vitamin E (alpha-tocopherol), beta-carotene and Superoxide dismutase will slow the process of aging by preventing free radicals from oxidizing sensitive biological molecules or reducing the formation of free radicals. The antioxidant chemicals found in many foods are frequently cited as the basis of claims for the benefits of a high intake of vegetables and fruits in the diet.Nonetheless, some recent studies tend to show that antioxidant therapy have no effect and can even increase mortality. Since many different substances operate synergistically in anti-oxidant defence, its complicated process may require more sophisticated approaches to determine if antioxidant therapy may benefit the aging process.
The Daily Omnivore
Everything is Interesting

Leave a comment